PET & SPECT
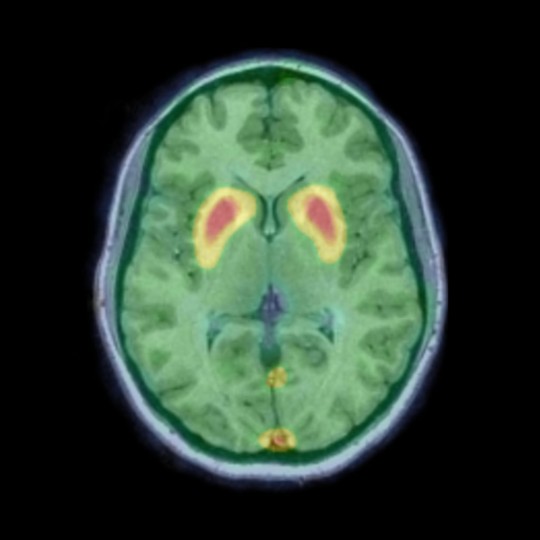
β-Amyloid PET
β-Amyloid plays a pivotal role in the pathobiology of Alzheimer’s disease and is the subject of research in understanding its exact role in neurodegenerative diseases as part of developing effective treatments against its adverse effects.
We provide fully automated SUVR (Standardised Uptake Value Ratio) and dynamic analysis methods for the quantification of amyloid pathology from PET imaging. Our method can utilise a subject-specific multi-atlas structural segmentation, allowing the flexibility to analyse multiple single and/or composite brain regions.
Our SUVR analysis has been applied to data collected as part of the Alzheimer’s disease Neuroimaging Initiative (ADNI) and Australian Imaging, Biomarker & Lifestyle Study of Ageing (AIBL) studies and validated using the Global Alzheimer’s Association Interactive Network (GAAIN) data to convert SUVRs to a standardised centiloid scale.
We are working with tracer manufacturers on a number of Alzheimer’s disease clinical trials and also as part of the Dementia Platform UK (DPUK) and AMYPAD projects – where we are analysing all 6,000 amyloid PET scans collected in the AMYPAD diagnostic and prognostic studies.
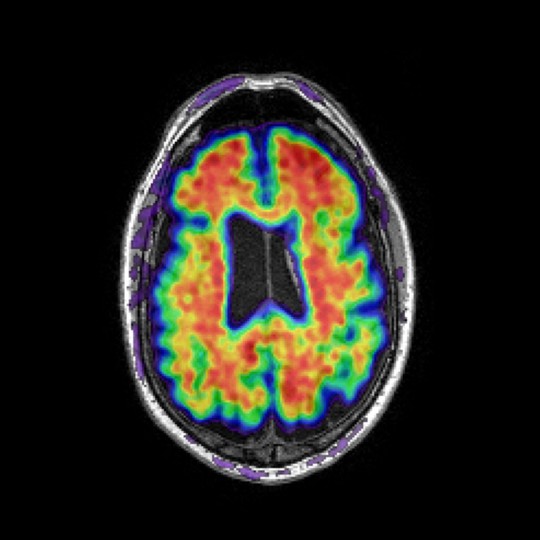
Tau PET
Tau proteins are associated with pathologies such as Alzheimer’s disease and Parkinson’s disease, where tau proteins have become defective and no longer stabilise microtubules properly, forming neurofibrillary tangles (NFT). The degree of NFT involvement in both diseases is defined by Braak stages – a system that characterises disease progression.
Our tau PET analysis allows us to quantify NFT distribution in different clinical groups, or monitor longitudinal changes. Among other regions, we can analyse the entorhinal cortex, hippocampus, amygdala, parahippocampus and fusiform – given that these regions better identify differences in NFT distribution in clinical populations and identify stereotypical pattern distribution in Alzheimer’s disease, according to successive Braak stages.
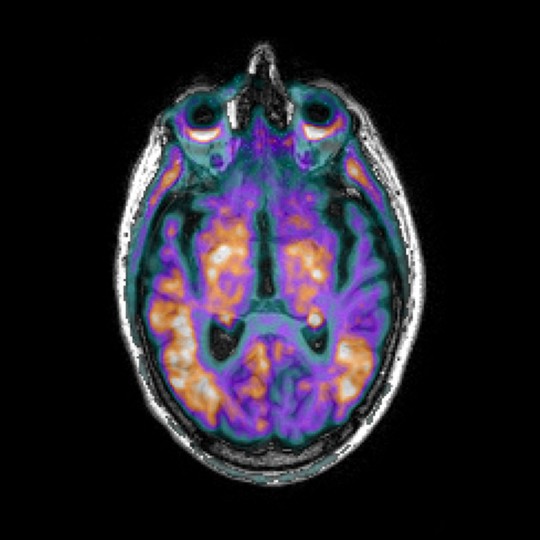
Flurorodeoxyglucose (FDG) PET
FDG PET imaging is used to observe metabolic processes in the body. FDG is an analogue of glucose, and its concentrations indicate metabolic activity as it corresponds to regional glucose uptake. As brain pathologies such as Alzheimer’s disease are characterised by decreased brain metabolism, FDG PET may be used to measure disease progression, or gauge the efficacy of new treatments longitudinally.
We use a fully automated SUVR method, which allows us to flexibly select multiple single and/or composite regions for the analysis. Our analysis has been successfully applied to PET scans from Phase II and Phase III Alzheimer’s disease clinical trials, and FDG PET images from the Alzheimer’s disease Neuroimaging Initiative (ADNI) and Australian Imaging, Biomarker & Lifestyle Study of Ageing (AIBL) studies.
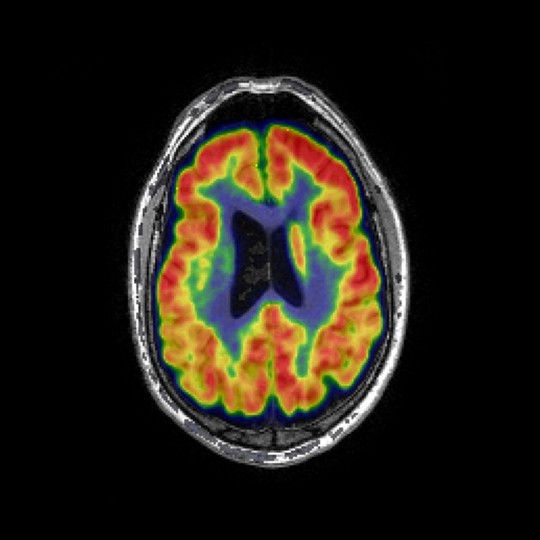
Dynamic PET
Dynamic scans allow for a more reliable quantification of specific imaging tracer binding by taking into account between-subject differences of tracer delivery and clearance rates. Different kinetic models can be used for different tracers to investigate several physiological processes. We provide the following kinetic models to analyse dynamic scans:
- Simplified Reference Tissue Model (SRTM) – to quantify nondisplaceable binding potential (dopamine receptor availability in Parkinson disease)
- Reference Logan – to quantify distribution volume ratio (amyloid and tau deposition in Alzheimer’s disease)
- Reference Patlak – to quantify tracer net uptake (dopamine uptake in Parkinson’s disease and FDG brain metabolism studies)